Hi Everyone,
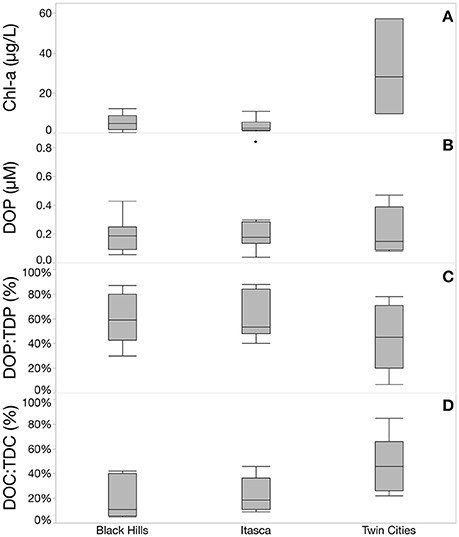
I have been sitting on the following scientific paper for a year and I suspect that it may be of interest to others for a variety of reasons. Ever wondered why algae grows in water in which there appears to be no measurable phosphate/phosphorus? Could it just be that the phosphate test kit is only able to detect inorganic phosphate - otherwise known as orthophosphate (PO4---)? Well, stay tuned - the following may be right up your street. Although its primary focus is temperate lakes, I suspect that the paper includes plenty of information that may be of interest to the aquatics hobbyist. Here it is:
 www.frontiersin.org
www.frontiersin.org
JPC
I have been sitting on the following scientific paper for a year and I suspect that it may be of interest to others for a variety of reasons. Ever wondered why algae grows in water in which there appears to be no measurable phosphate/phosphorus? Could it just be that the phosphate test kit is only able to detect inorganic phosphate - otherwise known as orthophosphate (PO4---)? Well, stay tuned - the following may be right up your street. Although its primary focus is temperate lakes, I suspect that the paper includes plenty of information that may be of interest to the aquatics hobbyist. Here it is:

Frontiers | Bioavailability of Dissolved Organic Phosphorus in Temperate Lakes
Freshwater aquatic systems are biogeochemical hotspots, with heterotrophic bacteria rapidly cycling the compounds that pass through them. P is a key nutrient...
JPC


