Courtneybst
Member
Hey everyone,
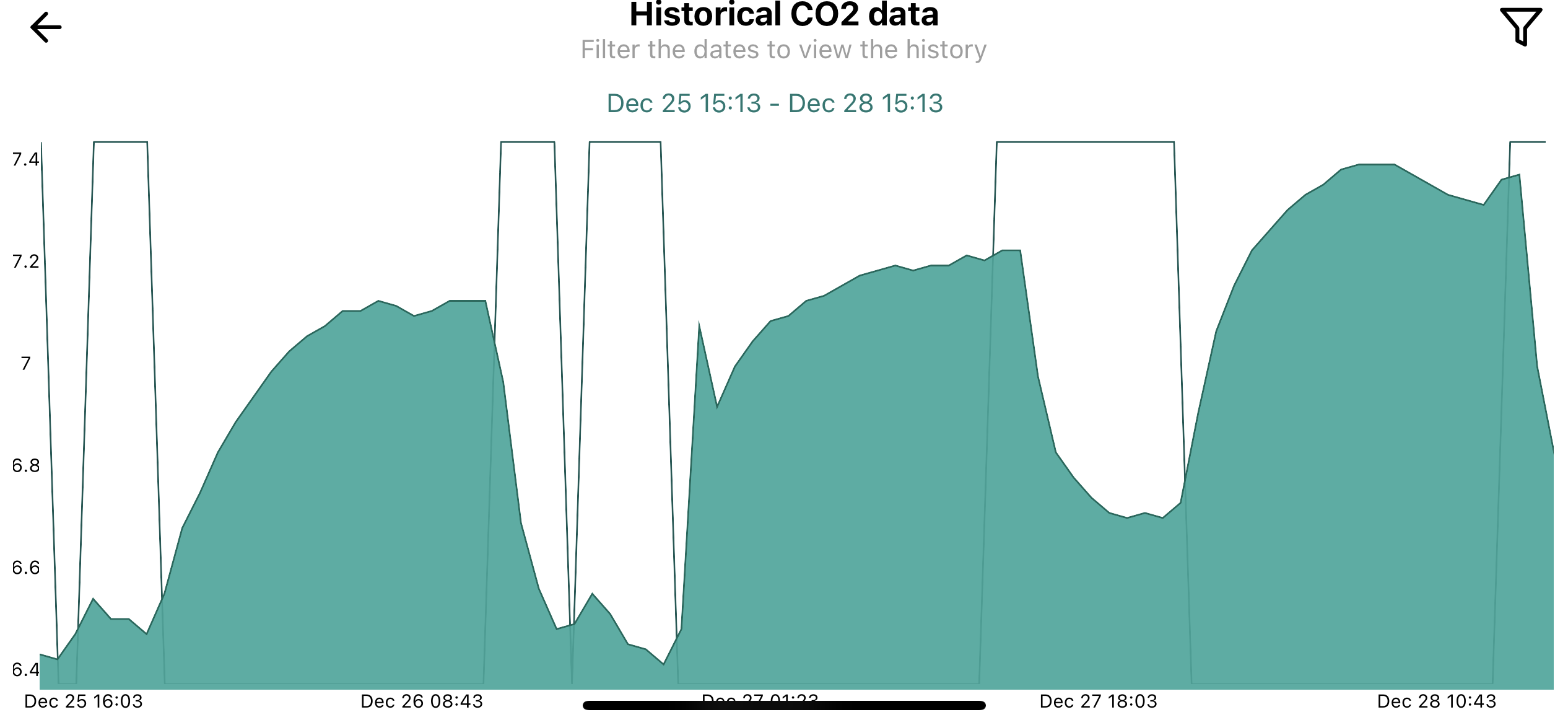
So after installing an Intaqo controller on my tank, it's provided me with some interesting data that I would have been none the wiser to.
My CO2 comes on 3 hours before the lights to try and achieve a 1 pH drop (or as close to). However, I've noticed that as the days progress since the last water change, there seems to be more and more resistance against the pH drop and the pH is increasing daily. I'm aware that some rocks influence pH slightly (I have Frodo/Millennium stone in this scape) but I just thought it was interesting.
Any ideas on whether this will negatively impact CO2 delivery? It's my understanding that in a higher pH you might never achieve a 1 pH drop so what is the acceptable drop in a higher pH? Is the pH drop completely irrelevant in this instance?
I've attached a graph from the last few days so you can see what I mean.
So after installing an Intaqo controller on my tank, it's provided me with some interesting data that I would have been none the wiser to.
My CO2 comes on 3 hours before the lights to try and achieve a 1 pH drop (or as close to). However, I've noticed that as the days progress since the last water change, there seems to be more and more resistance against the pH drop and the pH is increasing daily. I'm aware that some rocks influence pH slightly (I have Frodo/Millennium stone in this scape) but I just thought it was interesting.
Any ideas on whether this will negatively impact CO2 delivery? It's my understanding that in a higher pH you might never achieve a 1 pH drop so what is the acceptable drop in a higher pH? Is the pH drop completely irrelevant in this instance?
I've attached a graph from the last few days so you can see what I mean.

