-
You are viewing the forum as a Guest, please login (you can use your Facebook, Twitter, Google or Microsoft account to login) or register using this link: Log in or Sign Up
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
Combining chemical filtration media
- Thread starter sWozzAres
- Start date
Alastair
Member
I'd have thought when it came to changing the carbon, it would be a chore in itself seperating it out from the purigen and vice versa when it came to recharging the purigen
Sent from my iPhone using Tapatalk
Sent from my iPhone using Tapatalk
A
Antipofish
Guest
Alastair said:I'd have thought when it came to changing the carbon, it would be a chore in itself seperating it out from the purigen and vice versa when it came to recharging the purigen
Sent from my iPhone using Tapatalk
I agree, Alastair has a good point. Even if you intend chucking the purigen and adding new stuff, it lasts a lot longer than carbon which should be replaced every 4 weeks or so normally.
sounds like you've answered your own questions! If they were going to react, physical colocation in the same bag would make no difference - they would do so by dissolving in water, which they clearly dont.
One bag might improve flow especially if it meant 1/3 the amount of bag material vs media. In my experience these bags tend to be quite big and you end up with a fair bit scrunched up - that's defo not good for flow.
One bag might improve flow especially if it meant 1/3 the amount of bag material vs media. In my experience these bags tend to be quite big and you end up with a fair bit scrunched up - that's defo not good for flow.
A
Antipofish
Guest
what filter do you have, whats its media layout and how do you have your chemical bags set up ? If you have them so they dont block the whole flow portion of the media tray it will minimise flow restriction (in much the same way that JBL use a combi block in their filters where the outer foam allows good flow and the inner portion contains the chemical media. The flow through the chemical media will be slower than the surrounding foam, so your overall flow should not be affected too much whilst allowing the chemical media to still do its job.
Im curious why people have started to add carbon and phosphate remover?
I know Mark evans does on his current tank but said himself he was just trying it... we dose phosphates so why then remove it and the speculation that carbon removes goodness from the water that plants will use?
My 2 pence would be to take out the rowaphos and carbon, problem solved.
I know Mark evans does on his current tank but said himself he was just trying it... we dose phosphates so why then remove it and the speculation that carbon removes goodness from the water that plants will use?
My 2 pence would be to take out the rowaphos and carbon, problem solved.
A
Antipofish
Guest
easerthegeezer said:Im curious why people have started to add carbon and phosphate remover?
I know Mark evans does on his current tank but said himself he was just trying it... we dose phosphates so why then remove it and the speculation that carbon removes goodness from the water that plants will use?
My 2 pence would be to take out the rowaphos and carbon, problem solved.
Iain. Apparently (and Im no expert) there are phosphates and then there are PHOSPHATES. The ones we add do not get removed by the phosphate removers. Beyond that you will have to find the thread that covers it. Something to do with organics ? I was as confused as this reply sounds, but there is more to it than removing what we put in 🙂
Antipofish said:Iain. Apparently (and Im no expert) there are phosphates and then there are PHOSPHATES. The ones we add do not get removed by the phosphate removers. Beyond that you will have to find the thread that covers it. Something to do with organics ? I was as confused as this reply sounds, but there is more to it than removing what we put in 🙂
I have never heard of PHOSPHATES having a negative effect on a planted aquarium..?
Sounds like a marketing ploy to me
I would be interested to know what the science behind it is, if any.
A
Antipofish
Guest
easerthegeezer said:Antipofish said:Iain. Apparently (and Im no expert) there are phosphates and then there are PHOSPHATES. The ones we add do not get removed by the phosphate removers. Beyond that you will have to find the thread that covers it. Something to do with organics ? I was as confused as this reply sounds, but there is more to it than removing what we put in 🙂
I have never heard of PHOSPHATES having a negative effect on a planted aquarium..?
Sounds like a marketing ploy to me
I would be interested to know what the science behind it is, if any.
LOL. Like I said, there are phosphates and PHOSPHATES, I dont know either mate.
Pretty sure that in solution, all phosphate ions are equivalent. Phosphates coming from organic sources are still phosphates, but they may come with other less desirable organics.
Rowaphos is manly used in marine tanks to take po4 levels down to very low levels. Not sure if it will do much to drop phosphates from 3ppm. In any case, why bother? As You will read here, the mantra is "nutrients do not cause algae", and given the number of people dumping salts into their tanks via EI, they clearly don't bother fish at these levels. Save the money or just put more purigen in, as that removes the less attractive organics. And seriously, who can be ar*ed using a media that needs replacing every month?
Rowaphos is manly used in marine tanks to take po4 levels down to very low levels. Not sure if it will do much to drop phosphates from 3ppm. In any case, why bother? As You will read here, the mantra is "nutrients do not cause algae", and given the number of people dumping salts into their tanks via EI, they clearly don't bother fish at these levels. Save the money or just put more purigen in, as that removes the less attractive organics. And seriously, who can be ar*ed using a media that needs replacing every month?
Hi all,
cheers Darrel
Technically not quite, between pH5 - pH8 you mainly get a mix of HPO4-- and H2PO4- ions, below that pH you get H3PO4 and at high pH PO4--- . I don't think this makes any difference, and I would be fairly surprised if there is any validity inPretty sure that in solution, all phosphate ions are equivalent.
Without being overly cynical if I knew that phosphates were known to be a major cause of the eutrophication of fresh waters, even at extremely low levels, and almost impossible to test for without lab grade kit, I might be quite keen on selling a product that you can make all sorts of claims for, but that no-one can test.if Apparently (and Im no expert) there are phosphates and then there are PHOSPHATES. The ones we add do not get removed by the phosphate removers.
Agree, in fact I can't see the point of any chemical filtration media, unless there is a specific solute that needs removing (carbon after medication etc).And seriously, who can be ar*ed using a media that needs replacing every month?
cheers Darrel
actually i think the phosphate remover doubles as a silica remover thereby lowering the chance of diatoms during setup
anyway, 5 weeks into my new 350l and i have not had a diatom issue
only used the phosphate remover on startup
anyway, 5 weeks into my new 350l and i have not had a diatom issue
only used the phosphate remover on startup
Hi all,
A lot of us will have huge amounts of Diatom frustules in their tanks, the Moler clay in "Tesco's lightweight Cat litter" is diatomite, and comprises the "skeletal" remains of millions of Diatoms.
There is some more in this thread: <http://www.ukaps.org/forum/viewtopic.php?f=51&t=18067&p=185197>
If you really want to see "Chemical filtration and the matrix" in its full glory have a look at this video,
of the Rena API Nexx filter - <http://www.youtube.com/watch?v=ZvT1T77cjCg> and the "Rena Nexx Prevent Algae Filter Media" cartridges for it: <http://www.seapets.co.uk/products/a...event-algae-filter-media-size-4-two-pack.html>
I was actually asked to review this filter, but for some reason they haven't used my review " .....This filter is so laughably ill designed ....." or sent me one to do some road tests on.
cheers Darrel
If it is Rowaphos it would. This is another one I'm a bit agnostic about however. Diatoms can only incorporate orthosilicic acid (H4SiO4) to make their "skeletons" (Frustule), so silica is a bit of a red herring.actually i think the phosphate remover doubles as a silica remover
A lot of us will have huge amounts of Diatom frustules in their tanks, the Moler clay in "Tesco's lightweight Cat litter" is diatomite, and comprises the "skeletal" remains of millions of Diatoms.
There is some more in this thread: <http://www.ukaps.org/forum/viewtopic.php?f=51&t=18067&p=185197>
If you really want to see "Chemical filtration and the matrix" in its full glory have a look at this video,
of the Rena API Nexx filter - <http://www.youtube.com/watch?v=ZvT1T77cjCg> and the "Rena Nexx Prevent Algae Filter Media" cartridges for it: <http://www.seapets.co.uk/products/a...event-algae-filter-media-size-4-two-pack.html>
I was actually asked to review this filter, but for some reason they haven't used my review " .....This filter is so laughably ill designed ....." or sent me one to do some road tests on.
cheers Darrel
dw1305 said:If it is Rowaphos it would. This is another one I'm a bit agnostic about however. Diatoms can only incorporate orthosilicic acid (H4SiO4) to make their "skeletons" (Frustule), so silica is a bit of a red herring.
isn't orthosilicic acid just a dissolved form of silica, which as I understand isn't very soluble but when it does dissolve it can result in other silicic acids as well
Hi all,

From that impeccable source Wikipedia:
Your tank will always have enough silica to support diatom growth.

cheers Darrel
Yes, but silicic acids are formed by acidification of silicate salts (such as sodium silicate) in aqueous solution, not from the silicon (SiO2) in diatom shells. Diatom shells don't dissolve, they accumulate in huge amounts in sediments. This is the biogeochemical cycle for marine environments:orthosilicic acid just a dissolved form of silica, which as I understand isn't very soluble but when it does dissolve it can result in other silicic acids as well
From that impeccable source Wikipedia:
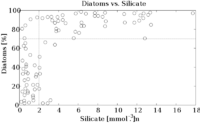
"......This family of compounds have the general formula [SiOx(OH)4-2x]n.[1][2] Some simple silicic acids have been identified, but only in very dilute aqueous solution, such as metasilicic acid (H2SiO3), orthosilicic acid (H4SiO4, pKa1=9.84, pKa2=13.2 at 25 °C), disilicic acid (H2Si2O5), and pyrosilicic acid (H6Si2O7);....."The use of silicon by diatoms is believed by many researchers to be the key to their ecological success. In a now classic study, Egge & Aksnes (1992)[16] found that diatom dominance of mesocosm communities was directly related to the availability of silicic acid — when concentrations were greater than 2 mmol m-3, they found that diatoms typically represented more than 70% of the phytoplankton community. Raven (1983)[17] noted that, relative to organic cell walls, silica frustules require less energy to synthesize (approximately 8% of a comparable organic wall), potentially a significant saving on the overall cell energy budget. Other researchers[18] have suggested that the biogenic silica in diatom cell walls acts as an effective pH buffering agent, facilitating the conversion of bicarbonate to dissolved CO2 (which is more readily assimilated). Notwithstanding the possible advantages conferred by silicon, diatoms typically have higher growth rates than other algae of a corresponding size.[12] Diatoms occur in virtually every environment that contains water. This includes not only oceans, seas, lakes and streams, but also soil.
Your tank will always have enough silica to support diatom growth.

cheers Darrel