Hi all, It is straightforward enough, but ppm TDS (parts per million Total Dissolved Solids) is a strange measurement.
To measure it accurately you have to filter the water and then evaporate a known weight of water to dryness. The water company will have done this, but usually ppm TDS is estimated from the electrical conductivity of the water.
If you took of a cup of sea-water, as it dried the sea salt (NaCl mainly), which had been in solution as ions (Na+ and Cl-), would crystallise out as the water (H2O) evaporated. The salt is a "dissolved solid".
It is the same process in fresh-water, but there
are a lot less salt(s).
If you evaporate de-ionised water (pure H2O) you wouldn't have any dissolved solids, and it wouldn't conduct electricity (it doesn't have any ions), but usually what we call "water" is a dilute solution, with water as the solvent. In sea-water we have a lot of ions (salts in solution) so it conducts electricity very well. The electrical conductivity of pure water is 0 microS and sea-water is 53,000 microS, and it is a linear scale between 0 and 53,000.
Conductivity measurements only measures ions, but normally they are the major contributors to TDS. You can have a dissolved non-ionic solid (like the dissolved sugar in your cup of tea, or the tannins from bog-wood) but, because most of the dissolved solids in water are ions, conductivity is reasonably well correlated with TDS.
If you have 100 microS conductivity that is equivalent to ~64 ppm TDS, assuming that Ca++ and HCO3- (from limestone CaCO3) are the major ions in fresh-water (and that is a pretty fair assumption).
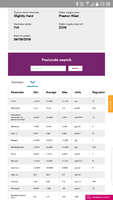
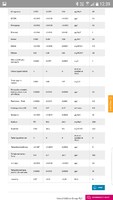
Your minimum, average and maximum figures for conductivity (in microS), in your water report, are 157, 250 and 405, which are approximately 100, 150 and 260 ppm TDS (157*0.64 = 100.5 etc.).
As
@Parablennius suggests that is a lot of variation in water hardness, so you have supplies from more than one source. Because you have virtually no nitrate in your supply the harder water is almost certainly from a deep limestone aquifer, and the softer water from a moorland reservoir.
cheers Darrel