In the meantime I'd like to ask some more questions if that's ok.
Of course! No problem at all.

You mention that reducing the photoperiod will help to battle the BBA problem whilst increasing intensity will help the plants. The latter I get, but why does a shorter photoperiod help with BBA? Why are we hurting BBA (or at least slowing it down?) by reducing the photoperiod whilst in the same time it helps the plants?
Fair question. This goes to the relative efficiency in which plants and algae use the different resources. As general rule, algae are better adapted to take advantage the low levels of CO2, for example, so when these levels are low, the plants are not having a good perform with the photosynthesis, which means they leave the terrain for algae, who can grow with lower concentration of CO2 and then take advantage of the nutrients the plants cannot control. However, under the right conditions, plants control better the environment. With the light a similar phenomena happens. A low PAR is associated to algae, because the plants are not so efficient. Algae behave as opportunist in terms of ecology, which means that they will always use the resources found in excess when others are missing. Aquatic plants, however, are specialists, so they perform better when the conditions are favoring them.
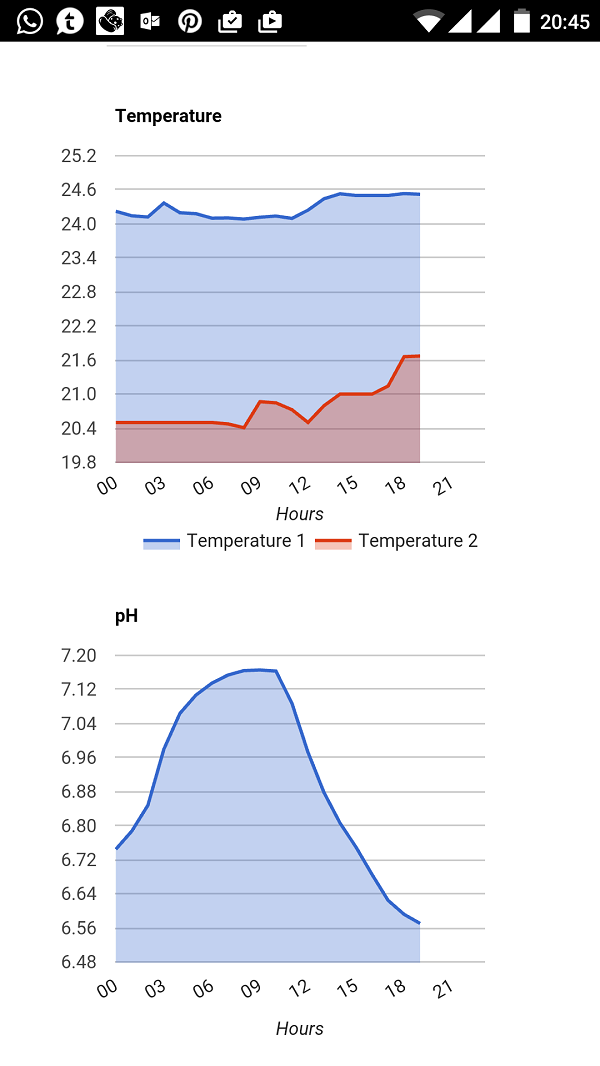
In the case I mentioned, having a low PAR for long hours will help algae. Plants cannot perform well with a low PAR because they will not getting enough energy for they photosynthetic processes. However, algae can use this reduced light, and as plants are not consuming nutrients, they have a based stone to build up. By increasing the intensity, you provide the right PAR to the plants, and under these conditions, plants will perform better and will use more the nutrients in water, which leaves no room for the algae. If the light is intense, plants do not need to get closer. The reason why your Staurogyne was growing tall is because the plant is trying to go closer to light to access to a better PAR. However, if the PAR is correct, the plant do not need this, so use its growth to cover the maximum horizontal surface as possible, as it is a better strategy for the plant if the light levels are good. However, as said before, algae will use what is in excess, so if you keep an intense light for long time and there are enough nutrients in water, then other types of algae will appear as these conditions can help them. In the case of BBA, it needs certain levels of light. I noticed that in my tanks never appear in areas that are dark. This is why when you were increasing your intensity, your BBA were happier. However, both plants and algae work in bases to the total amount of PAR received during a period of time. In other words: It is not only the PAR but also the time which gives the right dosage. But this is also on top of the fact that they need also a minimum intensity to work fine. With a low PAR for long hours what happens is that your intensity is not enough for the plants, but the accumulated radiation is enough for BBA, so essentially they have options to grow and compete with the plants.
In general, to ensure you beat algae, there are three rules that usually work: a) Reduce the amount of organic matter in decomposition (i.e. cleaning dead leaves, filter media, remove scum building up in the areas with low water flow); b) Reduce the photo-period to ensure algae cannot receive the PAR and dosage of radiation they need. Most plants survive some periods of time with no light at all, but many algae are not able to do so; c) Balance the nutrients (i.e. ensuring no nutrient is in deficiency) and reduce its dosage so we avoid to have any excess favoring opportunists (the commonly advised water changes goes in this direction).
I suspect I now also know why some of my helena snails have a battered house, it's probably because my rather high CO2 levels are consuming the carbonate in their houses, right?
Absolutely correct. The shells of molluscs are made of precipitated calcium carbonate and chitin (a kind of organic molecule used by many inverts to create hard structures). The CaCO3 is only stable in water if pH is about 7.8 or higher. This is due precisely to the equilibrium I mentioned above. When there are many protons in water, they will tend to bind to carbonates to produce bicarbonates, which are highly soluble in water. The calcium is also then released to the water. When you inject CO2 and your pH drops, then the shells of your snails will suffer the same acidic attack than carbonated rocks, so yes, that is why the appear battered. In fact, most of molluscs and crustaceans only appear in waters with high pHs, as otherwise they have problems to create/preserve their shells, which makes them more vulnerable to predators and diseases.
I was taught that in any tank, you want things to be as stable as possible so the plants (and fish) don't have to continuously adapt to changing parameters
Yes, totally right. You will find people telling that the aquatic organisms live in environments with a high variability of parameters, but that is quite false. Many of the species are very specialized to specific conditions (not all, of course), but despite of that, all species benefit of stable conditions. Jumping parameters just cause them problems and stress, which translates to a lack of growth and/or diseases.
What are the benefits of having a stable kH?
The main advantages are two: a) It buffers the water and stabilizes the pH so help a lot in the goal to keep a stable environment. pH jumps are cause of stress, and, opposite as said by some, natural waters keep a quite constant pH, unless we talk of some specific lakes which can have some annual cycle related to high productivity. Whwn we talk of rivers of springs, water parameters quite stable. So stable that some species of fishes like salmons are able to "smell" the water of the rivers by the signature they leave in the sea and go back to the place they were born. They could not do so if the water of these rivers were substantially changing; b) It helps to get the right levels of dissolved CO2 without having to drop too much the pH. Plants are more tolerant to low pH (not all of them) but even so, if the pH drops too much, they will be harmed by an acidic water. A kH between 3 and 8 is considered still very soft water but helps to avoid the pH having to drop too much to get the right CO2 levels.
If I want to stabilize the kH, it would be better to have some source of carbonates if I understand you correctly. Would that help in any way? Reason i ask is that I've kept a small bag of the old soil so I could place that in the tank or filter. That would even be controllable in the amount of carbonates it releases as I can add more soil or remove some when needed. The tap water I use has a kH of 9, so it can drop some points during the week to the point where I refresh the water again.
This is very difficult to achieve, and that is why many hobbyist just decide not to bother with kH, which eventually drops to zero if they do not have any source of carbonates in the tank, with a high risk of killing the fishes. Or maybe they have carbonates and happens the opposite if they do not compensate the increase of carbonates with water changes. So here, in your case, you had a very bad combination. kH of 9 is fine (a bit high for my taste but still good) in tap water. If you do not have any source of carbonates, the kH will drop due to CO2 injection, and will recover again when you change the water. However, depending of the drop of kH between water changes you suffer, and the amount of water you change, the recovery has some limits. For instance, in an extreme case, if your kH drops to zero between water changes and you change 50% of the water in your tank, after the maintenance you get a kH of 4.5, which will disappear sooner than when you started with 9, so your tank will spend more time at kH close to zero. On the other hand, if you have a kH of 9 but you have carbonated rocks, it could happen that you kH not only does not drop but that increases within the time, even with water changes: You start with 9 and between water changes increases up to 12. When you change 50%, after maintenance you kH goes to 10.5, higher than your starting kH of 9, so eventually increases to levels that are totally bad for your tank.
Finding the balance with the hardscape can be difficult, and the impact of rocks as seiryu depends on the amount you have. These rocks, however, are not so harmful as some sands, as the impact is also related to the total amount of surface of carbonated rocks in contact with water, which much much large in sands than in big rocks. Your idea of using some amounts of that sands to try to balance kH can help. Maybe best option is to introduce it in a net that you keep hide in the tank outside of sight or in the filter. You can play with the amount of that sand in the bag until you get a more stable kH. Note that, however, that kH will change anyway with the water changes, unless you aim to get it stable at the kH of your tap water.
In the case of my tank, kH slowly grows, but I compensate it from time to time by increasing CO2 levels and water changes.
The Akadama in it's first year would take down the kH to near 0 like some aquasoils do too. It's lost that property now it seems.
Yes, this is normal. Akadama and aquasoils make more acid the water by ion exchange, which is related to the composition of the clays that are their main mineral. They release protons to water usually as exchange of potassium. Eventually these exchange happens with most of the possible protons, so at the end the effect dissappears and they stop dropping the pH.
Hope this help.
Cheers,
Manuel